С помощью Скидосики, пользователи могут отслеживать снижение цен на интересующие их товары в различных
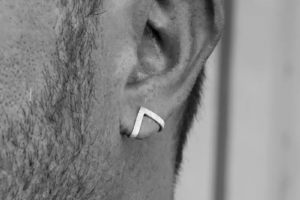
Сегодня мужские серьги становятся всё более популярными, и ассортимент предложений продолжает расти. Чтобы сделать
В этой статье мы рассмотрим ключевые аспекты и методы, которые помогут вам достичь идеальных
Малыши каждый год ждут подарки для детей новогодние сладкие. Вкусные наборы можно приобрести в
ИталКосметика - это итальянская компания, специализирующаяся на производстве профессиональной косметики для волос.
При выборе гель-краски рекомендуется обратиться к профессионалам или парикмахеру для получения советов и рекомендаций.
Аниматор - это специалист, который занимается развлекательной программой на мероприятиях, таких как детские праздники,
ПВХ (поливинилхлорид) мембрана - это вид материала, который широко используется в строительстве для создания
Зеркала с подсветкой - это популярное решение для ванных комнат, гримерных, гардеробных и других
Converse кроссовки - это обувь для спорта и повседневной носки, которая отличается от обычных
Если перманентный макияж выполнен не профессионально или не соответствует ожиданиям, его можно исправить с
Компания Lagom предлагает широкий ассортимент товаров для специалистов в сфере ресниц и бровей.
Домашние обеды - это готовые блюда, приготовленные дома или в небольших кулинарных предприятиях, и
Эксперименты с этими напитками все еще продолжаются. Однако есть одно правило, которое действует при
Когда вы ждете, пока воск погаснет, следует поместить его под картонную коробку или стеклянную
Если подложить под решетку ткань, сорная растительность не будет прорастать и ваши дорожки надолго
Модные оттенки помады в 2022 году, что будет в тренде в новом сезоне, новинки
Оригинальный запах с гармоничными нотками позволяет его обладательнице легко привлекать к себе внимание –
Продукция подлежит обязательной сертификации. Компания тщательно управляем контрактным производством во всех процессах, от сырья
Вибропрессованная тротуарная плитка – качественный и красивый материал для оформления дорожек, улиц, площадей, стоянок
Сухая и чувствительная кожа по мнению специалистов требует самого тщательного ухода. Многие женщины испытывают
Дано определение что такое бриллиант, приведены его основные характеристики. Даны подробные рекомендации как и
Магические, физические и лечебные свойства камня Шпинель. Кому подходит из знаков зодиака. Как правильно
Самые дорогие кольца в мире с уникальными бриллиантами идеальной чистоты продают на аукционах Sotheby's
Можно ли увеличить размер кольца с камнями Возникает ситуация, когда требуется увеличить размер кольца
Символизм кольца: в древнем мире, в Средневековье, в настоящее время. Какое значение вкладывалось в
Показано влияние колец с драгоценными камнями на красоту рук, подробно описаны их виды и
Мужские перстни, кольца и печатки дополняют гардероб, украшая ваш облик. Они запросто смогут разнообразить